ERS Charts of Note
Subscribe to get highlights from our current and past research, Monday through Friday, or see our privacy policy.
Get the latest charts via email, or on our mobile app for  and
and 
Monday, June 5, 2023
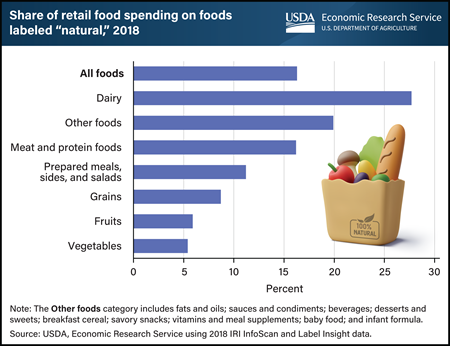
In 2018, food products labeled “natural” accounted for slightly more than 16 percent of all consumer retail food purchases. USDA and the U.S. Food and Drug Administration require producers to adhere to specific standards or processes to use certain label claims, such as USDA Organic. The “natural” claim, however, has minimal requirements and using the claim on a food product’s packaging does not require that the product provide any health or environmental benefits. Regulatory agencies treat the claim as meaning nothing artificial was added and the product was minimally processed. Even so, consumers sometimes attribute benefits to products labeled “natural,” research studies show. The share of products labeled “natural” varies by food category. The share of spending on “natural” products in 2018 was highest for dairy products (27.7 percent) and lowest for fruits (5.9 percent) and vegetables (5.4 percent). The data in this chart appear in the USDA, Economic Research Service report The Prevalence of the “Natural” Claim on Food Product Packaging, published in May 2023.
Friday, March 18, 2022
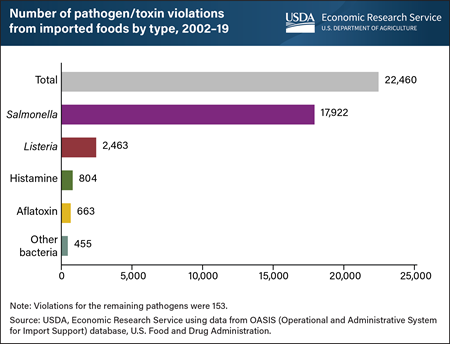
As the quantity of food imported into the United States continues to rise, it is increasingly important to minimize foodborne illness risks for U.S. consumers. Foods contaminated with pathogens or toxins can result in foodborne illnesses. A recent USDA, Economic Research Service (ERS) study examined the number of U.S. import refusals caused by pathogen/toxin contamination and which pathogens accounted for those safety violations. From 2002 to 2019, 22,460 pathogen/toxin violations were discovered among imported shipments. Salmonella was the most frequently identified agent among imported foods during the period with 80 percent, or 17,922 of total pathogen/toxin violations. Listeria recorded the second largest number of violations at 2,463, accounting for 11 percent of the total. It was followed by histamine with 804 violations (3.6 percent), aflatoxin with 663 violations (3 percent), and bacteria other than Salmonella or Listeria with 455 violations (2 percent). Those five most frequently detected pathogens and toxins accounted for 99.3 percent of the total pathogen/toxin violations from imported foods over the period. This chart was drawn from the ERS report Examining Pathogen-Based Import Refusals: Trends and Analysis From 2002 to 2019, published December 2021.
Monday, February 7, 2022
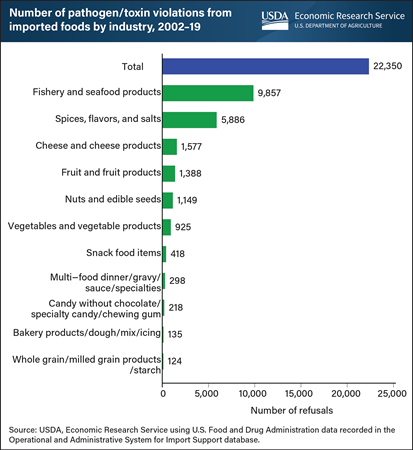
Food imported into the United States from other countries may contain pathogens such as bacteria, viruses, or other disease-causing microorganisms, or toxins, which are mostly produced by microorganisms. These pathogens and toxins could lead to foodborne illnesses. From 2002 to 2019, a total of 22,350 pathogen violations occurred from imported foods. About 70 percent of those violations came from two food sources: the fishery and seafood products industry and the spices, flavors, and salts industry. Fishery and seafood products had 9,857 pathogen violations over this period, accounting for 44.1 percent of the total refused imports. This category was followed by spices, flavors, and salts, which had 5,886 violations, or 26.3 percent of the total. Cheese and cheese products accounted for 7.1 percent of the total, followed by fruits and fruit products with 6.2 percent, nuts and edible seeds with 5.1 percent, and vegetables and vegetable products with 4.1 percent. In total, the top six food industries accounted for 93 percent of the total pathogen violations over the period. This chart was drawn from the USDA, Economic Research Service report Examining Pathogen-Based Import Refusals: Trends and Analysis From 2002 to 2019, published December 2021.
Tuesday, December 3, 2019
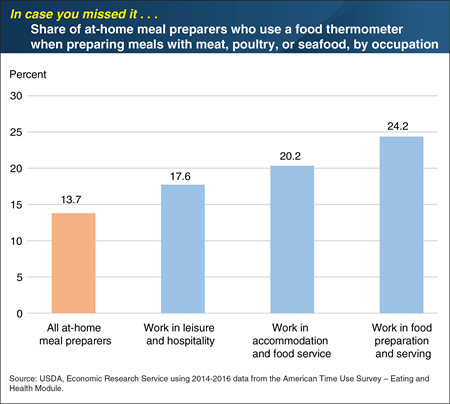
Public health officials recommend using food thermometers when cooking meat, poultry, and seafood to verify that the food is adequately cooked and that disease-causing pathogens, which could be in the food, are destroyed. Using data from the American Time Use Survey’s Eating and Health Module, ERS researchers found that 13.7 percent of at-home meal preparers used a food thermometer when preparing any meals with meat, poultry, or seafood during a typical week in 2014 to 2016. ERS researchers further examined whether at-home meal preparers with occupations related to food preparation were more likely to use a food thermometer. For at-home meal preparers who worked in the leisure and hospitality industry, 17.6 percent used food thermometers at home. Just over 20 percent of those who worked in the accommodation and foodservice industry, and 24.2 percent of those who worked in food preparation and serving occupations reported using food thermometers at home. These estimates exceed the national average, suggesting the training that foodservice employees receive for work partially carries over to behavior at home. This chart appears in the article, “Not All Consumers Are Following Food Safety Advice From Health Officials” in ERS’s Amber Waves magazine, April 2019. This Chart of Note was originally published May 22, 2019.
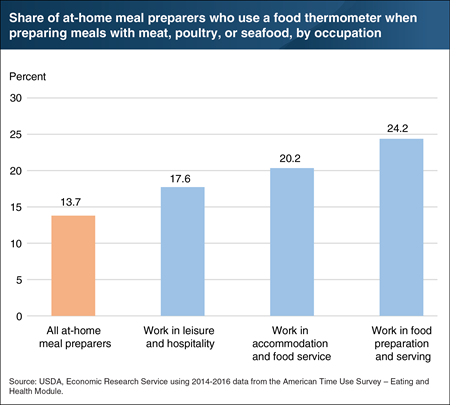
Wednesday, May 22, 2019
Public health officials recommend using food thermometers when cooking meat, poultry, and seafood to verify that the food is adequately cooked and that disease-causing pathogens, which could be in the food, are destroyed. Using data from the American Time Use Survey’s Eating and Health Module, ERS researchers found that 13.7 percent of at-home meal preparers used a food thermometer when preparing any meals with meat, poultry, or seafood during a typical week in 2014 to 2016. ERS researchers further examined whether at-home meal preparers with occupations related to food preparation were more likely to use a food thermometer. For at-home meal preparers who worked in the leisure and hospitality industry, 17.6 percent used food thermometers at home. Just over 20 percent of those who worked in the accommodation and foodservice industry, and 24.2 percent of those who worked in food preparation and serving occupations reported using food thermometers at home. These estimates exceed the national average, suggesting the training that foodservice employees receive for work partially carries over to behavior at home. This chart appears in the article, “Not All Consumers Are Following Food Safety Advice From Health Officials” in ERS’s Amber Waves magazine, April 2019.
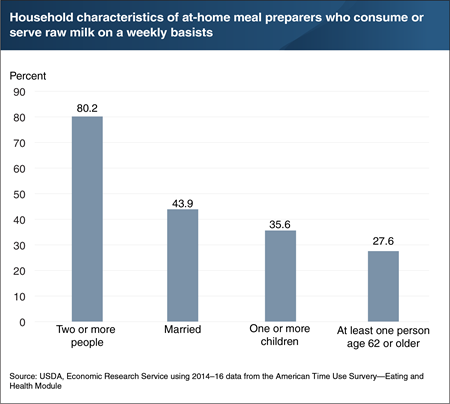
Tuesday, April 9, 2019
While pasteurization is required for interstate shipments of all milk and milk products intended for human consumption, many States allow intrastate shipments of raw (unpasteurized) milk from cows, sheep, or goats for human consumption. Raw milk can carry harmful bacteria and pose serious health risks, particularly for people with weakened immune systems, older adults, pregnant women, and children. From 2014 to 2016, an estimated 2.0 percent of at-home meal preparers, or 3.2 million people, consumed or served raw milk on a weekly basis. Many of these at-home meal preparers lived with one or more people in a high-risk group. Among at-home meal preparers who consumed or served raw milk, 80.2 percent lived with at least one other person, 43.9 percent were married, and 35.6 percent had at least one child under the age of 18 residing in the household. In addition, 27.6 percent had at least one person age 62 or older in the household. A version of this chart appears in the January 2019 ERS report, Consumer Food Safety Practices: Raw Milk Consumption and Food Thermometer Use. This topic is also discussed in the article, “Not All Consumers Are Following Food Safety Advice From Health Officials” in the April 2019 edition of Amber Waves.
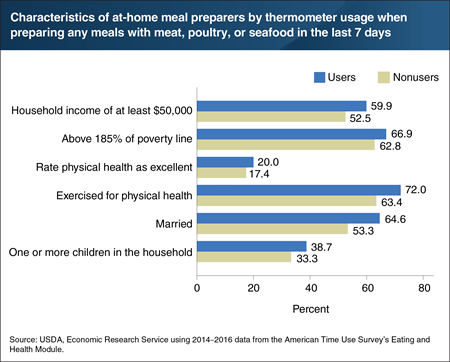
Tuesday, March 12, 2019
Improperly cooked meat can carry harmful bacteria and pose serious health risks, particularly for people with weakened immune systems, older adults, and children. Food thermometers are recommended when preparing raw meat to verify that food is adequately cooked and pathogens are destroyed. Using data from the American Time Use Survey’s Eating and Health Module, ERS researchers found that 14 percent of at-home meal preparers used a food thermometer during a typical week in 2014 to 2016. Households that used food thermometers were more likely to have an annual household income of $50,000 or more. Based on monthly pretax earnings and adjusting for household size, they were more likely to be above 185 percent of the Federal poverty line. At-home meal preparers who used a food thermometer were also more likely to rate their physical health as excellent, participate in physical activities or exercise for fitness in a given week, be married, and have at least one child in the household. A version of this chart appears in the ERS report Consumer Food Safety Practices: Raw Milk Consumption and Food Thermometer Use, January 2019.
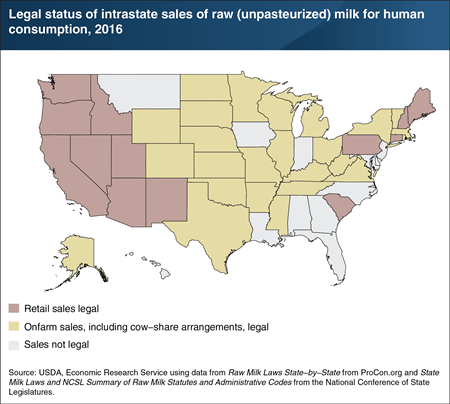
Wednesday, February 13, 2019
Public health authorities unequivocally advise consumers to avoid consuming raw (unpasteurized) milk. Raw milk can carry harmful bacteria and can pose serious health risks, particularly for people with weakened immune systems, older adults, pregnant women, and children. Pasteurizing milk—heating it for a specified period of time—kills dangerous bacteria and pathogens. Federal law requires pasteurization for interstate shipments of all milk and milk products intended for direct human consumption. However, States can allow intrastate shipments, and the number of States in which intrastate sale of raw milk from cows, sheep, or goats for human consumption is legal has been increasing. In 2016, 38 States allowed some form of intrastate sales of raw milk, 13 States allowed sales in retail stores, and 25 States allowed onfarm sales or cow-share agreements where a consumer can purchase a share of a cow’s milk production. Ten years earlier, intrastate sales in various forms were legal in 25 States. A version of this chart appears in the ERS report, Consumer Food Safety Practices: Raw Milk Consumption and Food Thermometer Use, released on January 29, 2019.
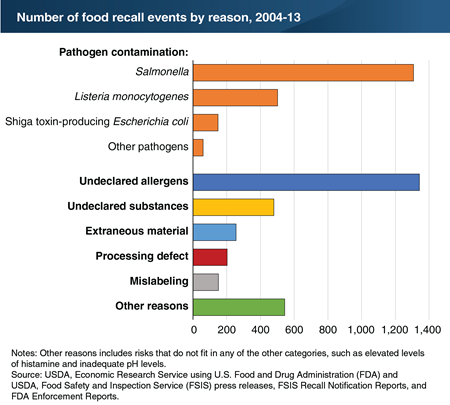
Thursday, July 12, 2018
Both USDA’s Food Safety and Inspection Service and the U.S. Food and Drug Administration oversee food product recalls—the removal of risky food products from the U.S. marketplace. From 2004 to 2013, there were 4,900 food recall events in the United States involving a wide variety of foods. Most of these recalls were initiated because of possible pathogen contamination (41 percent) and undeclared allergens (27 percent). Pathogen contamination is the discovery of disease-causing microorganisms, such as Salmonella and Listeria monocytogenes, in a food product. Undeclared allergens refers to the failure to declare on a food label one of eight major allergens: wheat, eggs, peanuts, milk, tree nuts (e.g., almonds, pecans, and walnuts), soybeans, fish, and crustacean shellfish. While the number of recalls due to pathogen contamination did not increase significantly from 2004 to 2013, the number of undeclared allergen recalls nearly doubled. The passage of the Food Allergen Labeling and Consumer Protection Act of 2004 (effective January 1, 2006) likely played a major role in the increase in the number of undeclared allergen recalls. The statistics for this chart are drawn from the April 2018 ERS report, Trends in Food Recalls: 2004-13.
Adulteration violations in imported foods increased the most for spices, flavors, salts, and seafood
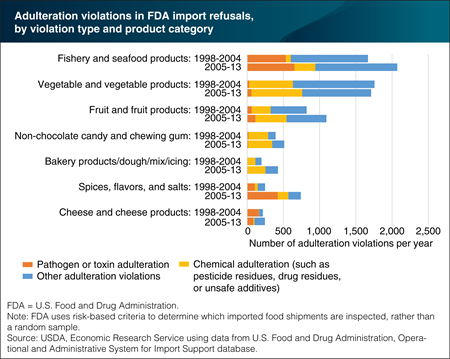
Friday, July 8, 2016
The U.S. Food and Drug Administration (FDA) oversees the safety of most food sold in the United States. Part of this oversight includes inspecting imported foods at the border or port of entry for evidence of adulteration or misbranding. FDA uses risk-based criteria to determine which shipments are inspected, rather than a random sample. A recent ERS study examined patterns in FDA import refusals over 2005-2013 and compared results with an earlier study of data from 1998-2004. Compared with the earlier period, spices, flavors, and salts, as well as fishery and seafood products, had the largest increases in the number of violations per year for adulteration—problems relating to safety issues, packaging integrity, or sanitation. In fishery and seafood products, the most common adulteration violations were for filth (visually apparent non-food material), the presence of Salmonella bacteria, and veterinary drug residues. In spices, flavors, and salts, the most common violation was for Salmonella. This chart is from “Patterns in FDA Imported Food Refusals Highlight Most Frequently Detected Problems” in ERS’s Amber Waves magazine, March 2016.
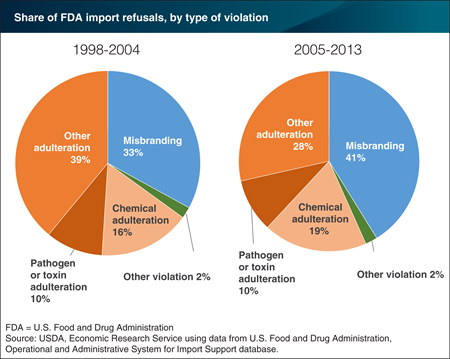
Thursday, March 31, 2016
The U.S. Food and Drug Administration (FDA) is responsible for overseeing the safety of most food sold in the United States, including food imported from other countries. A recent ERS study examined patterns in FDA import refusals over 2005-13 and compared results with an earlier study of data from 1998-2004. In both time periods, the majority of violations were for adulteration—problems relating to poisonous ingredients, disease-causing bacteria and viruses (pathogens), unsafe color additives, pesticide residues, or filth (visually apparent non-food material). Two product groups—fishery/seafood products and spices/flavors/salts—were responsible for the majority of violations for Salmonella bacteria. Chemical adulteration, including pesticide residues, accounted for a slightly larger share of import refusals in 2005-13. Chemical adulteration is a common type of adulteration violation in fresh produce and fruit and vegetable products. Misbranding violations for false, misleading, or missing labels accounted for 41 percent of violations in 2005-13, up from 33 percent in 1998-2004. The data for this chart come from the ERS report, FDA Refusals of Imported Food Products by Country and Category, 2005–2013, released on March 28, 2016.
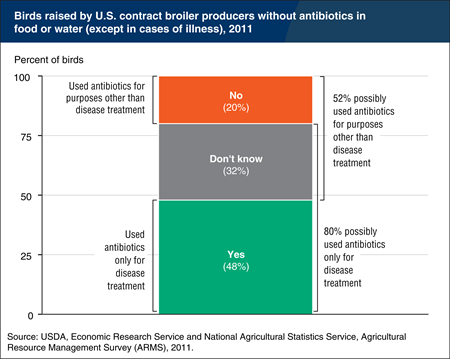
Thursday, March 17, 2016
Livestock farmers use antibiotics to treat, control, and prevent disease, and also for production purposes, such as increasing growth and feed efficiency. A new U.S. Food and Drug Administration initiative seeks to eliminate the use of medically important antibiotics for production purposes. In the 2011 Agricultural Resource Management Survey (ARMS) on broilers (the most recent year available), producers were asked whether they raised their broilers without antibiotics in their feed or water unless the birds were sick, which implies not using antibiotics for growth promotion or disease prevention. In 2011, growers reported that about half of birds (48 percent) were only given antibiotics for disease treatment. This response also accounts for 48 percent of operations and 48 percent of production (by live weight). Approximately a third (32 percent) of operators stated that they did not know if they provided antibiotics via feed or water for purposes other than disease treatment; this means the proportion of reporting operations that only supplied antibiotics for disease-treatment purposes could be as high as 80 percent. Contracted growers (accounting for 96 percent of broiler production) may not know if antibiotics are in the feed provided by the company for whom they raise broilers. These statistics suggest that in 2011, between 20 and 52 percent of birds were given antibiotics for reasons other than disease treatment. This chart is found in the Amber Waves feature, “Restrictions on Antibiotic Use for Production Purposes in U.S. Livestock Industries Likely To Have Small Effects on Prices and Quantities,” November 2015.
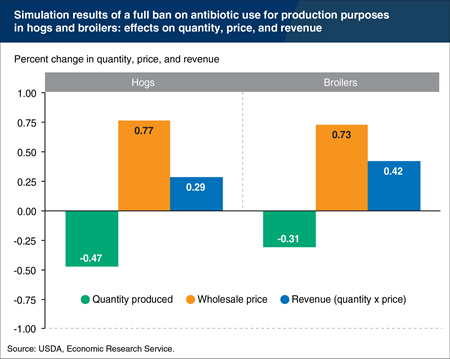
Tuesday, February 2, 2016
Livestock farmers use antibiotics to treat, control, and prevent disease, and also for production purposes, such as increasing growth and feed efficiency. A new U.S. Food and Drug Administration initiative seeks to eliminate the use of medically important antibiotics for production purposes. ERS research shows that only a portion of hog and broiler producers use antibiotics for production purposes, and the productivity increases from such uses are 1-3 percent. Modelling the effect of production-specific antibiotic restrictions suggests that such a policy would have a modest effect on wholesale prices and quantities produced of chicken and pork—less than a 1-percent increase in wholesale prices and a net decline in production of less than 0.5 percent. Because prices increase more than quantities decrease, gross revenues (price times quantity) would increase slightly. This chart is based on the table found in the Amber Waves feature, “Restrictions on Antibiotic Use for Production Purposes in U.S. Livestock Industries Likely To Have Small Effects on Prices and Quantities,” November 2015.
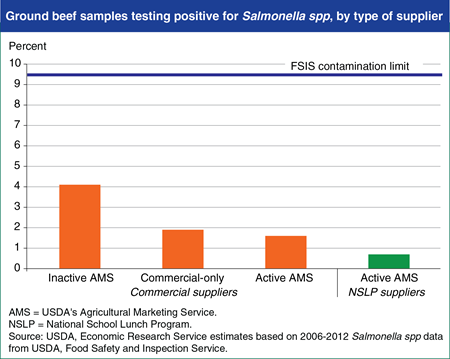
Friday, February 27, 2015
USDA’s Agricultural Marketing Service (AMS) provides about half of the ground beef served in the National School Lunch Program (NSLP) by purchasing raw and cooked ground beef products from U.S. meat producers. AMS-approved suppliers must meet AMS’s basic standards. AMS suppliers can be “active” and sell to the NSLP or be “inactive” and sell in commercial markets only. Active ground beef suppliers must adhere to a strict Salmonella standard. A recent ERS study found that on average, ground beef from all groups of producers examined had levels of Salmonella below the limit set by USDA’s Food Safety and Inspection Service. Researchers also found that AMS standards incentivize producers to supply ground beef to the NSLP that has lower levels of Salmonella contamination (0.7 percent of samples testing positive) than the ground beef the same suppliers sell to commercial buyers (1.6 percent of samples testing positive). Plants supplying ground beef to the NSLP performed better on Salmonella tests than commercial-only and inactive AMS plants. This chart appears in “Strict Standards Nearly Eliminate Salmonella From Ground Beef Supplied to Schools” in the February 2015 issue of ERS’s Amber Waves magazine.


